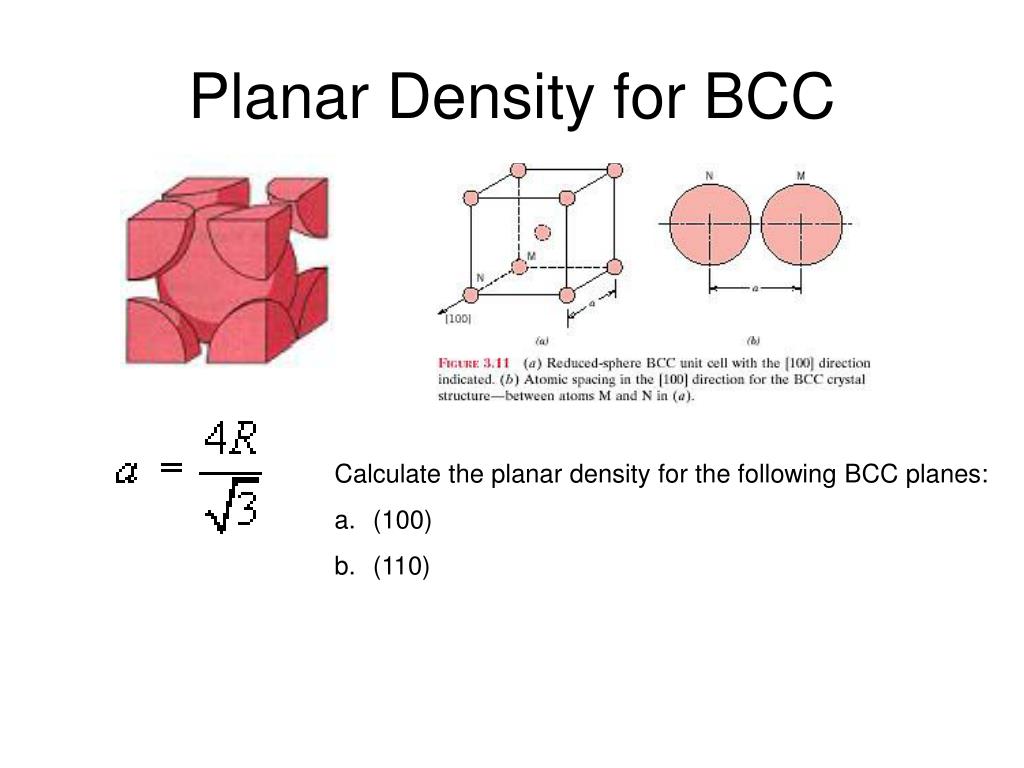
Exam 376 .pdf - Question: Sketch the ion arrangement of the 110 plane of ZnS with the zincblende structure and compare this arrangement to that on | Course Hero

Linear Density, Planar Density, Atomic Packing Factor {Texas A&M: Intro to Materials (MSEN 201)} - YouTube

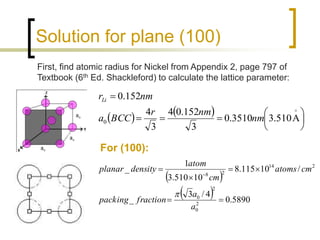
SOLVED: Determine the planar density and planar packing factor on the plane of a BCC crystal with a lattice parameter of 0.2460 nm. Planar density (three decimals): atom/cm^2 (Format: x.xxxE+xx atom/cm^2) Planar
What is Atomic Packing Factor (and How to Calculate it for SC, BCC, FCC, and HCP)? – Materials Science & Engineering
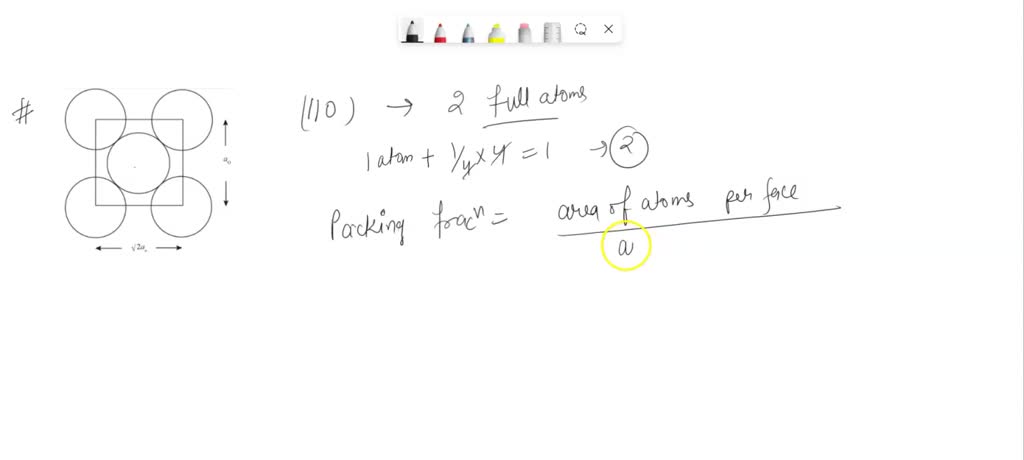
SOLVED: What is the planar packing fraction for the ( -110) plane in a BCC structure? A diagram is necessary as part of your calculation. Show all work.
![SOLVED: For aluminum at 300K, calculate the planar packing fraction (fractional area occupied by atoms) of the (110) plane (15 marks) and the linear packing density (atoms/cm) of the [100] direction (15 marks). SOLVED: For aluminum at 300K, calculate the planar packing fraction (fractional area occupied by atoms) of the (110) plane (15 marks) and the linear packing density (atoms/cm) of the [100] direction (15 marks).](https://cdn.numerade.com/ask_images/f329ab1695d54a948db52d0a81ec707c.jpg)
SOLVED: For aluminum at 300K, calculate the planar packing fraction (fractional area occupied by atoms) of the (110) plane (15 marks) and the linear packing density (atoms/cm) of the [100] direction (15 marks).

Solved) - 1. Determine the planar density and packing fraction for FCC... - (1 Answer) | Transtutors